Hydrogen Cells
Hydrogen fuel cells can be used in a wide range of applications such as transportation, portable generators, and emergency backup power supplies. Fuel cells have more benefits over conventional combustion-based technologies that many power plants and vehicles are using today. Hydrogen fuel cells are able to operate at higher efficiencies than combustion engines, and are able to convert the chemical energy in the fuel into electrical energy with the efficiency of up to 60%. Since hydrogen fuel cells emit water, there would be no carbon dioxide emissions and no air pollutants that create smog and cause health problems in the environment.
Solar Panels
Solar energy does not emit any green house gases since it uses the sun as the power source. Solar energy doesn't require the expensive and ongoing raw materials like oil or coal. It also offers decentralization in most locations that has sun, which means that societies are able to become self reliant societies. In order to work solar panels collect solar radiation from the sun and actively convert that energy to electricity. When the solar cells are then exposed to sunlight, the p-n junction diodes in the solar cell then convert the sunlight energy into usable electric energy.
Series and parallel circuits
A series circuit is a circuit where all of the resistances are in a row and that there is only one path for the electricity to flow. A application for a series circuit would be a string of light bulbs. If one light bulb blew, the remaining light bulbs would turn off since the path for the electricity to flow was broken which won't allow the electric current to flow. A parallel circuit is a circuit that has two or more paths for the electricity to flow and the resistances are parallel to each other in the circuit. If the light bulb in a parallel circuit blew the current would continue to flow to other bulbs.
Reflection
I think that we did a ok job as a group when we were building the car. As a team I believe that we could have helped each other out more on the project and calculations. There were multiple distractions that I noticed that greatly impacted the way that the team interacted with each other on the project. However we did get the car done and working but unfortunately we were not able to complete the calculations even with a extended deadline. It was also hard to find parts that I needed since the boxes containing the parts were not equally balanced with the same parts but that factor is not controllable. In the end I think that without the distractions effecting the team we could have finished the project and calculations and have time for improvements on the car to make it better and faster then it was before.
Typed Activity 1.3.1
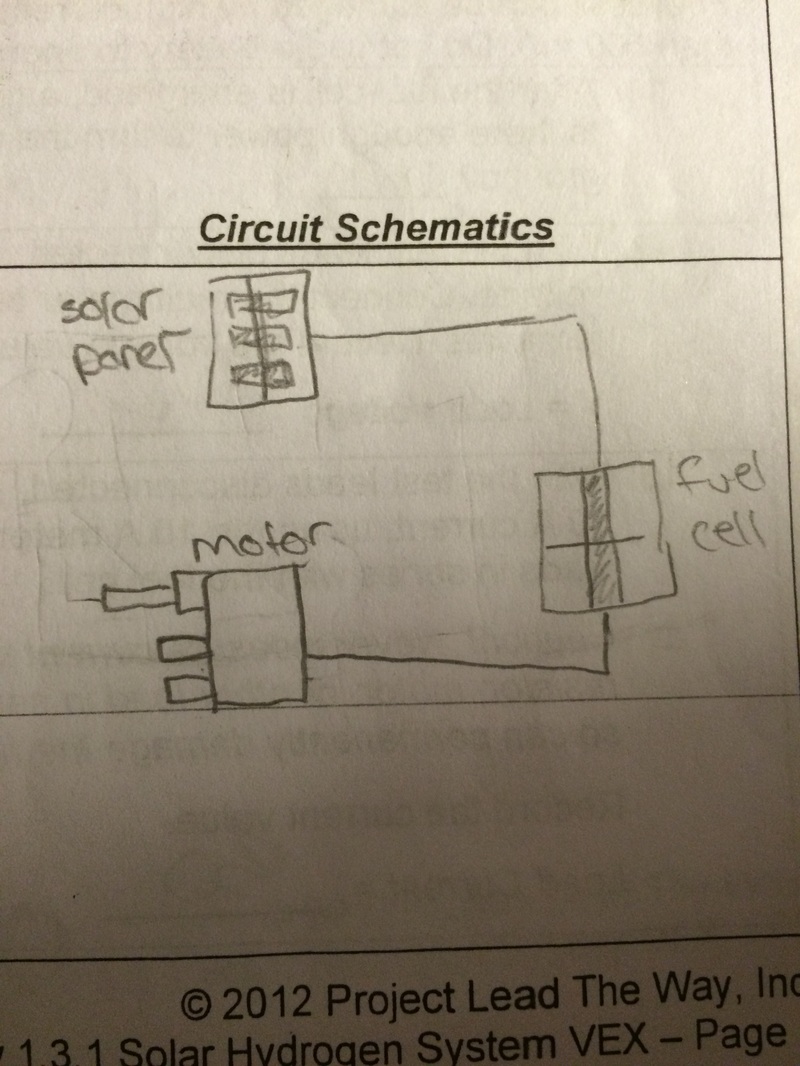
In activity 1.3.1 we had to design, build, test, and race a car that would run on a hydrogen fuel cell. We would use vex parts to build the car, a light and solar panel to charge the hydrogen fuel cell, a motor to move the car that was built, and a electric board/mother board to control the electric current from the hydrogen fuel cell to the motor. We would then have to test the open-circuit voltage, load voltage, short circuit current, load current, maximum theoretical power, and load power. After doing the calculations we would then draw circuit schematics for the relating problem and calculation.
Conclusion Questions
1) From what we got tested the fuel cell was better then what we got for the solar module since for the solar module we got 340.38 mW which is .340 watts (in the picture above it says 340.38 Watts which is wrong) and for the fuel cell we got 6 Watts which is a lot better then the solar module.
2) Power best described the suitability of the tested power sources.
3) Since we did not test the AAA batteries on our car I would not know the exact amount of solar modules need but since it didn't perform as well as the fuel cell, by estimate I would say it would need at least 10 to 12 solar modules to get the same performance of two AAA batteries.
4) Same as in question number 3 I did not get to test the AAA batteries but I would assume that 2 fuel cells can match the same performance of two AAA batteries.
5) I think that a parallel circuit system connected to multiple hydrogen fuel cells would meet the needs of an average driver since the fuel cells would be able to move the car and if one fuel cell stops working the car can still run due to the parallel circuit that allows the electric current to keep flowing.
6) Photovoltaics is the direct conversion of light energy into electric energy at the atomic level. Some materials exhibit a property called the photoelectric effect that causes those materials to absorb photons of light and release electrons. When the released electrons are captured, an electric current is produced and that current can be used as electric energy. http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7) Electrolysis is a method of separating elements by pushing a electric current through the existing compound. As the electricity from the power source passes through the electrodes in the water the water splits into hydrogen and oxygen and the hydrogen collects around the cathode while the oxygen collects around the anode. http://www.energyquest.ca.gov/projects/split_h2o.html
2) Power best described the suitability of the tested power sources.
3) Since we did not test the AAA batteries on our car I would not know the exact amount of solar modules need but since it didn't perform as well as the fuel cell, by estimate I would say it would need at least 10 to 12 solar modules to get the same performance of two AAA batteries.
4) Same as in question number 3 I did not get to test the AAA batteries but I would assume that 2 fuel cells can match the same performance of two AAA batteries.
5) I think that a parallel circuit system connected to multiple hydrogen fuel cells would meet the needs of an average driver since the fuel cells would be able to move the car and if one fuel cell stops working the car can still run due to the parallel circuit that allows the electric current to keep flowing.
6) Photovoltaics is the direct conversion of light energy into electric energy at the atomic level. Some materials exhibit a property called the photoelectric effect that causes those materials to absorb photons of light and release electrons. When the released electrons are captured, an electric current is produced and that current can be used as electric energy. http://science.nasa.gov/science-news/science-at-nasa/2002/solarcells/
7) Electrolysis is a method of separating elements by pushing a electric current through the existing compound. As the electricity from the power source passes through the electrodes in the water the water splits into hydrogen and oxygen and the hydrogen collects around the cathode while the oxygen collects around the anode. http://www.energyquest.ca.gov/projects/split_h2o.html